Lab
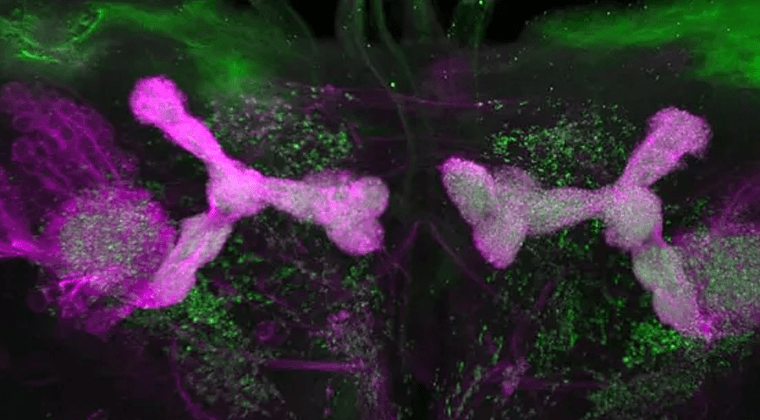
Ackerman Lab
The Ackerman lab uses both zebrafish and fruit fly model systems to determine how distinct glial cell populations (individually and collectively) instruct nervous system development, from synapses to circuits.

Cashikar Lab
Oxysterols are hydroxylated cholesterols, which influence a wide variety of cellular pathways including endolysosomal trafficking, cholesterol biosynthesis and cell surface and nuclear receptor signaling among others. These intracellular effects manifest as major regulatory steps in lipid homeostasis, inflammatory signaling, neuromodulation and cell migration. We are interested in understanding how oxysterols influence neuroinflammation, synaptic dysfunction and neurodegeneration in Alzheimer’s disease.
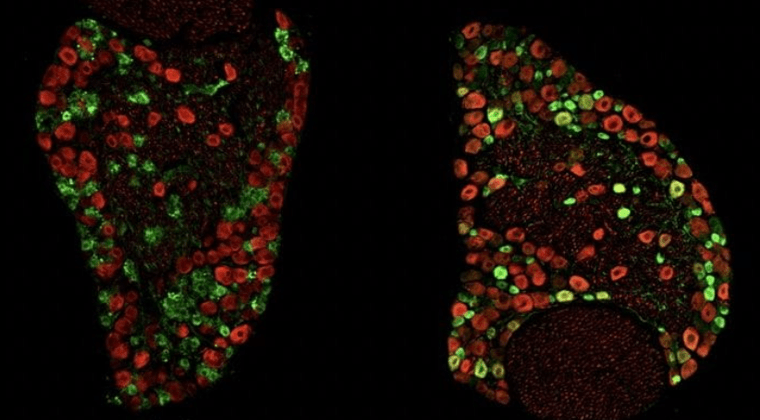
Cavalli Lab
Research in Cavalli laboratory focuses on elucidating the neuronal, glial and immune mechanisms that dictate the regenerative response of peripheral sensory neurons and to relate this information to the lack of regenerative capacity in the central nervous system.
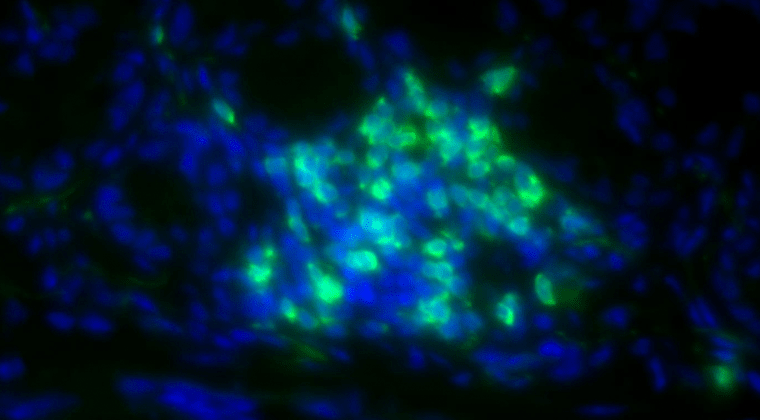
Colonna Lab
The laboratory is broadly interested in innate immunity, focused on three main areas:
1) Innate lymphoid cells (ILCs) in mucosal immunity
2) Innate immune mechanisms in Alzheimer`s disease and neurodegeneration
3) Plasmacytoid dendritic cells and IFNα/β in host defense and autoimmunity
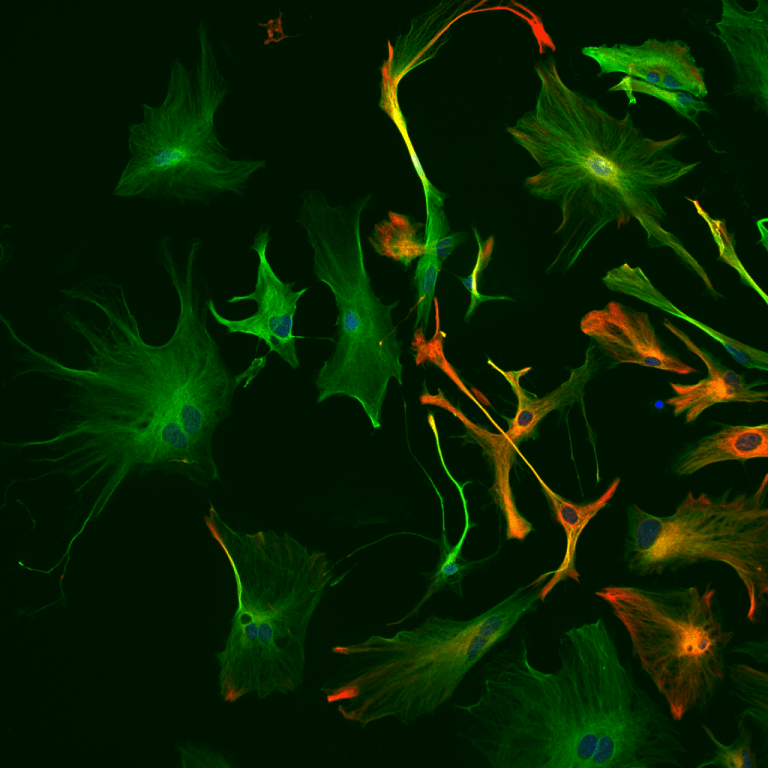
Cooper Lab
We are investigating the disease mechanisms that underlie lysosomal storage disorders, and use this information to devise new experimental therapies. This work focusses upon the Neuronal Ceroid Lipofuscinoses (NCLs, or Batten disease). These are profoundly neurodegenerative disorders that kill children, and most attention has so far focused upon neuron loss. However, we have discovered that both astrocytes and microglia are functionally compromised in these disorders and may play an active role in promoting neurodegeneration. We are now looking at the roles of glia, especially early in disease and how this differs between forms of NCL.

Cross Lab
The goal of our research is to understand the mechanisms involved in pathogenesis of inflammation and demyelination in the central nervous system (brain and spinal cord). Our research is primarily focused on the human disease multiple sclerosis (MS), which is very common (1/1000 in the US population). We study human tissues (blood, DNA, RNA, spinal fluid) and perform cutting-edge imaging of the human nervous system. We sometimes employ an animal model for MS known as experimental autoimmune encephalomyelitis (EAE). We are currently performing longitudinal imaging studies to help devise methods to differentiate the various types of pathologies in the human disease, such as demyelination, inflammation with cells and or edema due to blood-brain barrier breakdown, and axon damage and loss. We are funded to devise better methods to measure progression of MS over time, which might be used in clinical trials. We also study the effects of diet and calorie restriction upon neuro-inflammation, having shown previously that calorie restriction ameliorates EAE, the prime animal model for MS, at least partly through the upregulation of adiponectin and downregulation of leptin.
PI: Anne Cross, MD

Cruchaga Lab
My research interests are focused on leveraging multi-omic data (genetic, genomics, proteomics, epigenomics, lipidomis and others) and deep clinical phenotypes from large and well characterized neurodegenerative diseases (i.e: Alzheimer, Parkinson, Frontotemporal dementia) cohorts in order to identify novel genes, pathways, molecular biomarkers and drug targets for these diseases.

Davis Lab
Dr. Davis’ laboratory research is aimed at understanding the molecular and cellular mechanisms of protein aggregation and neurodegeneration in Parkinson Disease, with a special interest in factors that contribute to dementia in Parkinson Disease and related disorders including Dementia with Lewy Bodies.
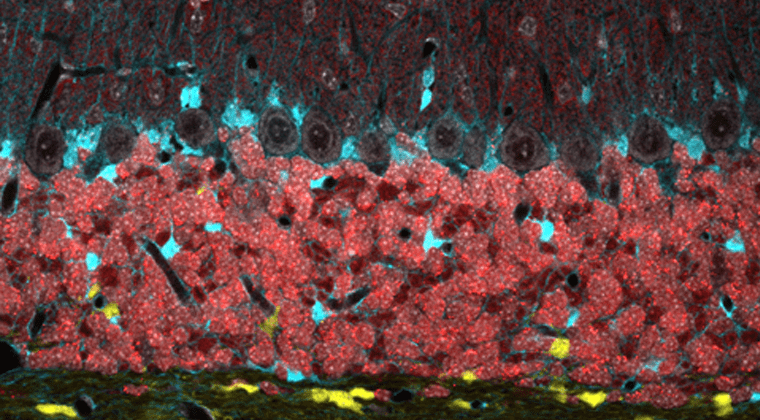
Dougherty Lab
The Dougherty lab develops novel neurotechnology to better understand transcriptional and translational regulation in health and disease, with a focus on astroglia.
Gallardo Lab
Our lab is interested in studying astrocytes as they are beginning to emerge as a critical component of Alzheimer’s disease (AD). Our goal is to understand the mechanisms that regulate reactive astrocytes by utilizing a combination of biochemistry, molecular biology, cellular models of inflammation, and mouse models of AD.
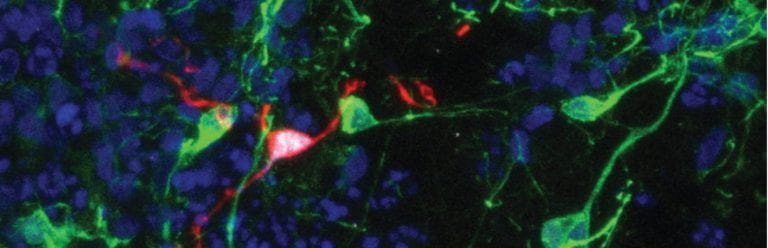
Han Lab
The Han lab seeks to elucidate the mechanisms by which microglia and other brain macrophages contribute to the neurodevelopmental and neurodegenerative disorders, through the lens of epigenetics and transcriptomics.
PI: Claudia Han, PhD

Hershey Lab
The Hershey lab works in the fields of cognitive and clinical neuroscience, using a range of neuroimaging, pharmacological and cognitive techniques to understand the impact of metabolic (e.g. obesity and diabetes) and neurodegenerative conditions on the brain, particularly during development. We draw from numerous disciplines (neuroscience, radiology, endocrinology, neurology, psychiatry, psychology) to enhance the quality and clinical relevance of our research.
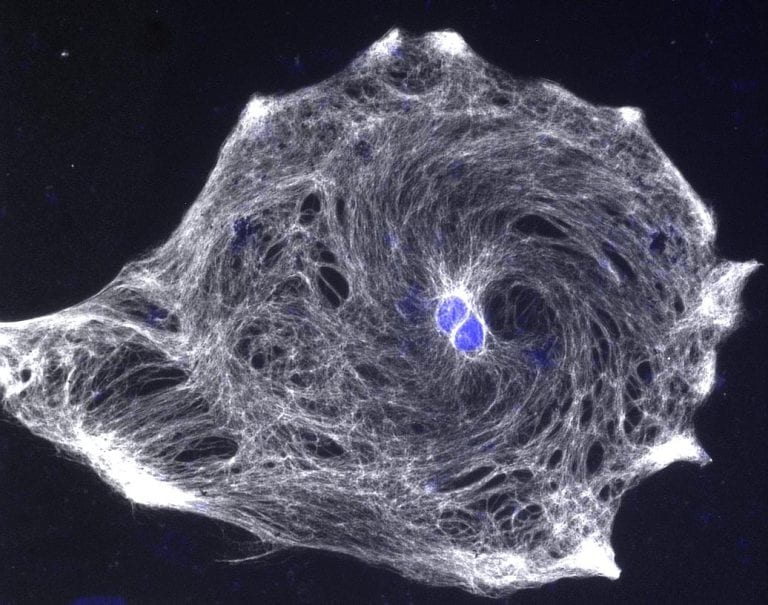
Herzog Lab
Astrocyte regulation of daily behaviors.
PI: Erik Herzog, PhD
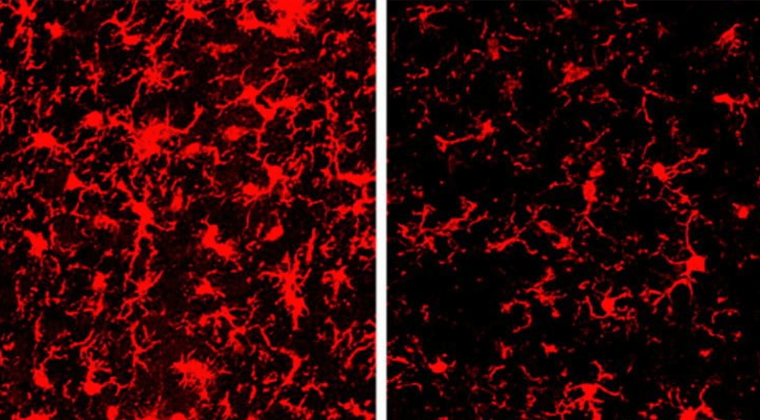
Holtzman Lab
The lab aims to better understand mechanisms underlying neurodegeneration, particularly as they are relevant to Alzheimer’s disease (AD). The lab has also been studying tau metabolism and how anti-tau antibodies may affect its potential role in spreading in its pathophysiology as well as how apoE influences tau-mediated neurodegeneration. The lab has also been studying how microglia and specific microglial genes such as TREM2 influence neurodegeneration in the setting of Aβ and tau pathology.
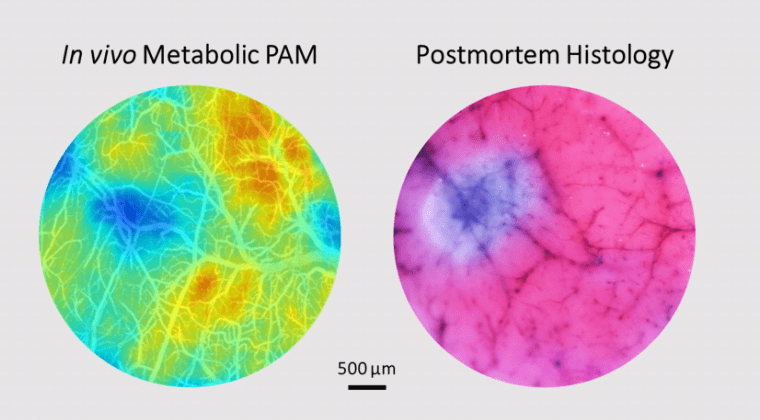
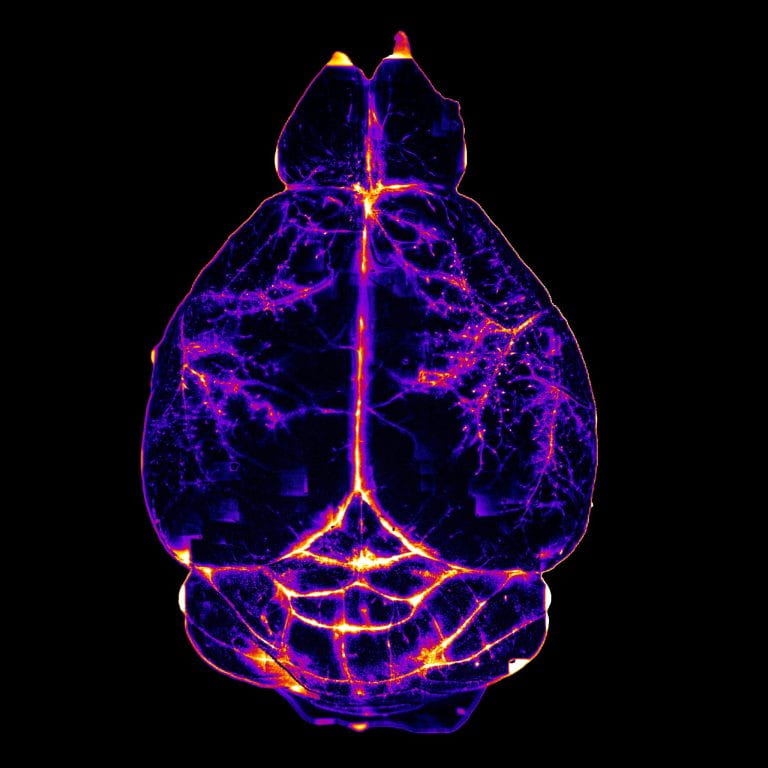
Hu Lab
Our lab research is centered on the development of cutting-edge optical and photoacoustic technologies for high-resolution structural, functional, metabolic, and molecular imaging in vivo and their applications in both basic and translational brain research.
PI: Song Hu, PhD
Kipnis Lab
We study complex interactions between the immune and nervous systems. Simply put, we investigate how the nervous and immune systems talk to each other in health and disease. We have discovered lymphatic vessels in the tissues surrounding the brain, a finding that has challenged some of the previous dogmas in the field of neuroimmunology and increased our knowledge and understanding of how the immune system impacts neurological diseases.
Kummer Lab
Research in the Kummer lab is focused on the mechanisms of cellular damage in traumatic brain injury and in Alzheimer’s disease, with a particular focus on synaptic and other forms of gray matter injury.

Lee Lab
We are engaged in translational research to investigate cellular and molecular mechanisms involved in acute and chronic brain injury, with a focus on Ischemic stroke and Alzheimer’s disease.
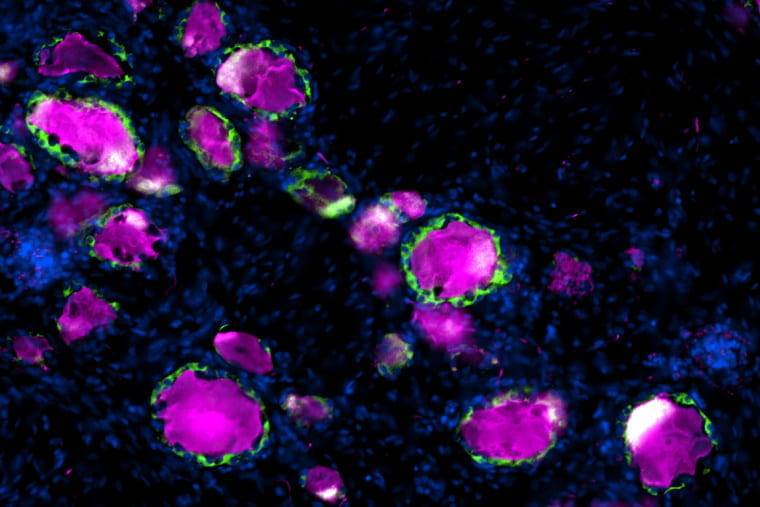
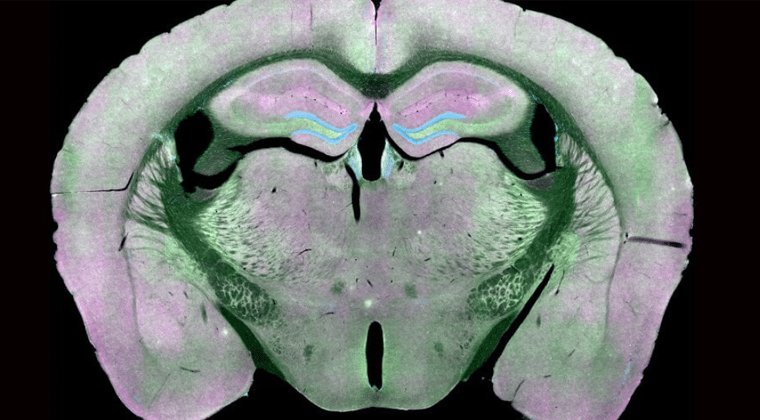
Li Lab
Microglia and other related immune cells are increasingly recognized to be essential players in central nervous system function and almost all neurological diseases. As we gain more knowledge about microglial biology—one of the fastest growing fields at the nexus of neuroscience, immunology and hematopoiesis, numerous fundamental questions remain to be answered, such as: How is microglia fate specified? How do microglia develop? How heterogeneous are microglia? How do microglia age? How do microglia contribute to brain development, aging and neurodegenerative disease? My lab is broadly interested in neuroimmunology with a focus on microglial biology. We combine cutting-edge single-cell genomic technologies with in vitro and in vivo genetic, molecular and cellular tools to address these fundamental questions, which also have tremendous translational potential. The overarching goal is to gain a better understanding of microglial functions in the establishment of the nervous system, as well as how changes in these functions contribute to neurological diseases.
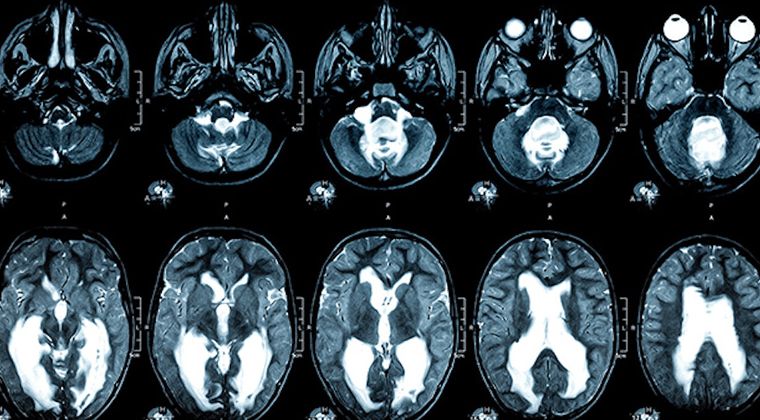
Limbrick Lab
Our laboratory focuses on cerebrospinal fluid physiology, both under normal conditions and in pathological states such as hydrocephalus and syringomyelia. We are particularly interested in the biology of the ventricular zone—the wall of the ventricle—and the subjacent subventricular zone, and the effect of injury to these areas on brain development.